Thoracic aortic aneurysms are much less common than abdominal aortic aneurysms. And, to top it off, the measurements of the thoracic aorta are different than the abdominal aorta, especially in the ascending thoracic aorta. It’s good to know how to look at the thoracic aorta when you need to, and what the normal measurements are so that you can make that immediate decision when a thoracic aneurysm (or dissection) is detected.
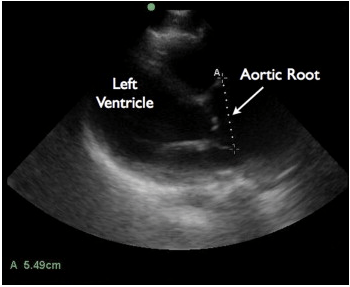
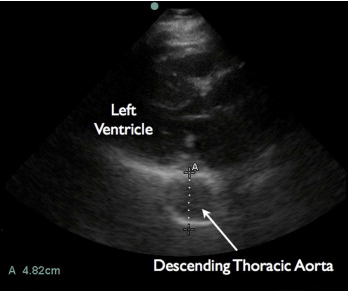
Drs. Daignault, Saul, and Lewiss published 2 great case reports in the Journal of EM Aug 2013 issue (subscription needed) on thoracic aortic aneurysms. One patient was a 60 year old male who had blunt trauma to his back by a heavy piece of plaster while at work. He had a BP of 140/80 mm Hg and HR of 90 beats/min. The FAST was negative for free fluid but the aortic root was found to be 5.49cm. This caused them to order a CT angio confirming their findings. Another patient was an 82 year old male with 1 month of chest pain radiating to his back with history of aortic valve repair and HTN on coumadin. BP was 210/90 mm Hg. A bedside echo was performed and showed descending thoracic aorta aneurysm measuring 4.82cm. This had a CT Angio done confirming descending thoracic aneurysm. The abstract is found here, and a prior post on these case reports can be found here.
These cases bring up an excellent reason to review the thoracic aorta. The best cardiac echo view to see visualize the thoracic aorta, if you could only pick one, would be the parasternal long view.
According to a german study where they looked at the thoracic aorta throughout life from 17 to 89 yrs of age via helical CT with the following methods and results: “Methods: Seventy adults, 17 to 89 years old, without any signs of cardiovascular disease were investigated with helical computed tomography. Aortic diameters were measured at seven predefined thoracic levels. Results: Aortic diameters (mean ± SD) were 2.98 ± 0.46 cm at the aortic valve sinus, 3.09 ± 0.41 cm at the ascending aorta, 2.94 ± 0.42 cm proximal to the innominate artery, 2.77 ± 0.37 cm at the proximal transverse arch, 2.61 ± 0.41 cm at the distal transverse arch, 2.47 ± 0.40 cm at the isthmus, and 2.43 ± 0.35 cm at the diaphragm. Men had slightly longer diameters than did women. All diameters increased with age. There was no influence of weight, height, or body surface area.”
More recently, Medscape authors state that the thoracic aorta dimensions are larger than the abdominal aorta dimensions and that aneursymal definition occurs when it is greater than 50% of the normal size.
Another study in the radiology literature with many more subjects more recently, had the following methods and results: “Methods: 1442 consecutive subjects who were referred for evaluation of possible coronary artery disease underwent coronary CT angiography (CTA) and coronary artery calcium scanning (CACS) (55+11 years, 65% male) without known coronary heart disease, hypertension, chronic pulmonary and renal disease, diabetes and severe aortic calcification. The ascending aortic diameter, descending aortic diameter (DAOD), pulmonary artery (PAD) and chest anterioposterior diameter (CAPD), posterior border of sternal bone to anterior border of spine, were measured at the slice level of mid right pulmonary artery by using end systolic trigger image. The volume of four chambers, ejection fraction of left ventricle, and cardiac output were measured in 56% of the patients. Patients demographic information, age, gender, weight, height and body surface area (BSA), were recorded. The mean value and age specific and gender adjusted upper normal limits (mean + 2 standard deviations) were calculated. The linear correlation analysis was done between AAOD and all parameters. The reproducibility, wall thickness and difference between end systole and diastole were calculated. Result: AAOD has significant linear association with age, gender, descending aortic diameter and pulmonary artery diameter (P<0.05). There is no significant correlation between AAOD and body surface area, four chamber volume, LVEF, CO and CAPD. The mean Intra-luminal AAOD was 31.1 ± 3.9mm and 33.6 ± 4.1 mm in females and males respectively. The upper normal limits (mean + 2 standard deviations) of Intra-luminal AAOD, mean+ standard deviation, was 35.6, 38.3 and 40 mm for females and 37.8, 40.5 and 42.6 mm for males in age group 20 to 40, 41 to 60, above 60 year respectively. Intra-luminal should parallel echocardiography and invasive angiography. Traditional cross sectional imaging (with computed tomography and magnetic resonance imaging) includes the vessel wall. The mean total AAOD was 33.5mm and 36.0 mm in females and males respectively. The upper normal limits (mean + 2 standard deviations) of Intra-luminal AAOD, mean+ standard deviation, was 38.0, 40.7 and 42.4 mm for females and 40.2, 42.9 and 45.0 mm for males in age group 20 to 40, 41 to 60, above 60 year respectively. The inter and intra observer, scanner and repeated measurement variability was low (R value >0.91, P<0.001, coefficient variation <3.2%). AAOD was 1.7 mm less in end-diastole than end systole(P<0.001).” Below is their table illustrating the various measurements by others:

As Dr Lewiss and colleagues state in their case reports, “The most recent consensus statement by the American Society of Echocardiography (ASE) and the American College of Emergency Physicians (ACEP) recognized that the thoracic aortic pathology can be identified on EP-performed focused cardiac ultrasound (3). Transthoracic echocardiography was shown to be consistent with TEE for measurement of the ascending aorta (21). Taylor et al. demonstrated that EP-performed focused cardiac ultrasound was consistent with CTA measurements for maximal thoracic aortic diameter (2). In these cases, an EP with considerable experience in ultrasound performed these studies, but many EP are trained in the use of focused ultrasound for evaluation of the abdominal aorta. Principals used in this application, such as avoiding measurements in oblique planes, measuring from outer wall to outer wall (for the descending thoracic aorta), and the use of Doppler also may be utilized in the evaluation of the thoracic portion of the aorta.”