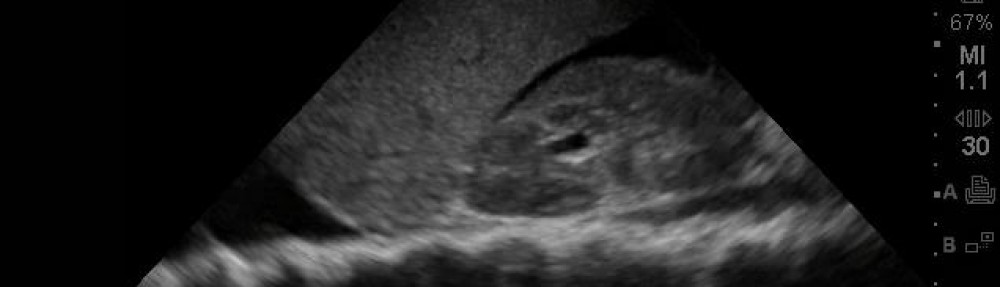
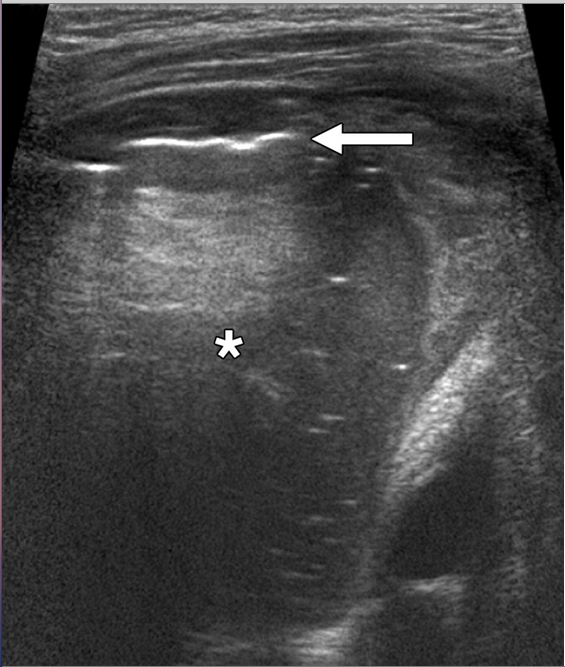
In a recent Medscape article, a study is highlighted regarding serial DVT ultrasound (including doppler of iliacs) can rule out DVT in pregnancy. How about that?! This is a follow up to that last post regarding “best test” for first diagnosis of suspected DVT. The article is stated below:
“Conducting serial compression ultrasonography with Doppler imaging of the iliac vein over the course of 7 days can rule out deep-vein thrombosis (DVT) in symptomatic pregnant women, according to a prospective cohort study.
Wee-Shian Chan, MD, from the Department of Medicine, BC Women’s Hospital and Health Centre, Vancouver, British Columbia, Canada, and colleagues report their findings in an article published online January 14 in the Canadian Medical Association Journal.
Increasingly, healthcare providers suspecting DVT turn to serial venous compression ultrasonography to diagnose the blood clots; if negative, Doppler imaging is added to overcome ultrasound’s lack of sensitivity in diagnosing isolated iliac DVTs in pregnant women for whom pelvic veins cannot be compressed. The diagnostic approach is noninvasive, widely available, and does not expose the fetus to ionizing radiation, and thus has become the standard of practice for diagnosing DVT in pregnant women. Because no study has validated this approach, Dr. Chan and colleagues sought to evaluate the diagnostic accuracy of using serial compression ultrasonography and Doppler imaging of the iliac veins among symptomatic pregnant women during a 7-day period.
They focused on pregnant women consecutively referred for investigation of DVT from August 2002 to September 2010 at 4 Canadian centers and enrolled 221 women who had had a suspected first episode of DVT. Exclusion criteria included receipt of anticoagulant therapy for more than 48 hours before presentation, suspicion of pulmonary embolism, concomitant cardiac or pulmonary disease, and inability or lack of willingness to return for follow-up.
Trained technicians performed ultrasound examination of the symptomatic leg by compression of the proximal veins (ie, along the length of the femoral vein from the inguinal canal and the popliteal veins to the level of the calf trifurcation with Doppler studies of the iliac vein). If the first test results were negative for DVT, blood thinners were withheld and testing was repeated after 2 to 4 days and again after 6 to 8 days. Blood thinners were prescribed if results were positive. The research team excluded DVT based on full compressibility of the femoral and popliteal veins and normal Doppler imaging of the iliac veins.
“The overall prevalence of deep vein thrombosis in our study cohort was 7.7% (95% [confidence interval,] 4.9% – 12.0%),” Dr. Chan and colleagues write. “The iliac or femoral veins, or both, were involved in 65% (11/17) of cases. Of these, 2 (12%) cases were isolated to the iliac vein, and 4 (24%) were isolated to the femoral vein.”
Some 94% of DVT were detected by the initial compression ultrasonography with Doppler imaging. The serial testing that followed did not detect any new thromboses, the research team writes. “Our strategy of serial compression ultrasonography combined with Doppler imaging of the iliac veins appears to reliably exclude clinically important deep vein thrombosis,” the authors write.
Added Value Unclear
Additional studies are needed to determine what is better for pregnant women: ultrasonic testing with Doppler imaging or single whole-leg ultrasound, “the only other diagnostic strategy that has been formally validated for DVT suspicion in pregnancy,” Grégoire Le Gal, MD, from the University of Brest, France, told Medscape Medical News. Dr. Le Gal and colleagues found that conducting a single ultrasound test may rule out diagnosis of DVT in pregnant and postpartum women.
“In nonpregnant patients, the 2 strategies have been compared and showed similar safety. The use of single whole-leg ultrasound is convenient, because patients don’t need to come back for serial testing. But, on the other hand, it is more time-consuming and it leads to a positive diagnosis of DVT in a higher proportion of patients, mainly because all distal DVT are detected — and therefore treated — with anticoagulants, when this approach is used,” Dr. Le Gal says. Using serial compression ultrasonic testing, “only the distal DVT that extend proximally are detected on serial tests and treated.”
Dr. Le Gal also noted that in Dr. Chan’s study, a Doppler interrogation was performed in all patients. “Physicians willing to implement this diagnostic strategy in their clinical practice need to take this into account. Unfortunately, the yield of this test was not provided in the manuscript. Therefore, it is impossible to know the exact added value of this test as compared with a serial compression ultrasonography without Doppler,” Dr. Le Gal told Medscape Medical News.
Because it is unethical to use leg venography with fluoroscopy or computed tomographic angiography in pregnant women to confirm DVT diagnoses, Dr. Chan and colleagues assumed that all diagnosed abnormalities were DVT. Another study limitation was that the current study showed a lower prevalence of DVT compared with 2 previous studies involving pregnant women; this could exaggerate the negative predictive value. In addition, the study investigators were thrombosis specialists based in secondary and tertiary referral centers, which raises the question of generalizability; because patients were recruited from a range of settings, however, the researchers argue that the study’s results are generalizable to most community-based hospitals.
“Our study highlights the importance of iliac vein visualization in symptomatic pregnant women,” the authors conclude. They add that the research also “shows that serial compression ultrasonographic studies with iliac vein visualization performed over a 7-day period can reliably exclude deep vein thrombosis in symptomatic pregnant women and that it is likely safe to withhold anticoagulation in women with negative imaging results.”
CMAJ. Published online January 14, 2013. Full text